
If the electrolyte level drops, add only distilled water. Don't overfill. If this still happened or a little electrolyte spilled out, immediately wipe it off and wash the area with water and soda. the electrolyte is very aggressive, in contact with the metal, the latter is highly corroded.
In addition to keeping the battery contacts clean and coated with petroleum jelly, the top cover of the battery, especially the top of the cell jars, must be kept clean and dry. This prevents corrosion and partial self-discharge of the battery through moisture and dirt.
Every three months, remove the battery and check the condition of its mounting bolts, mounting plates, pan and wires for corrosion (white fluffy deposit on metal, brittle to the touch). If corrosion is found, clean deposits with ammonia and coat the cleaned metal with an anti-rust agent or primer.
If there is excess buildup on the top of the battery then this could be caused by cracks in the case which cause electrolyte leaks or overcharging of the battery, indicating that the voltage regulator needs to be checked by a workshop.
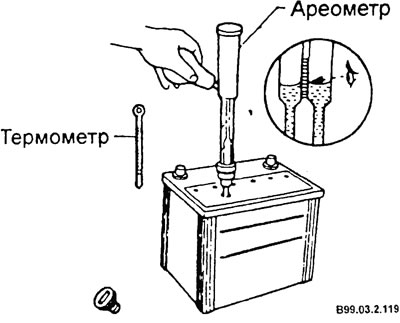
When checking the battery, once every three months, check the density of the electrolyte using a hydrometer to determine the state of charge of the battery and the state of the electrolyte.
- Type the electrolyte into the hydrometer through a special hole on the battery case. At the same time, measure the temperature of the electrolyte. If the electrolyte level is low, tilt the battery for easier measurement.
- Convert the density of the electrolyte at a given temperature to the density at a standard temperature of 20°C.
Example: electrolyte temperature 35°C measured density 1.230. The reduced density will be 1.240.
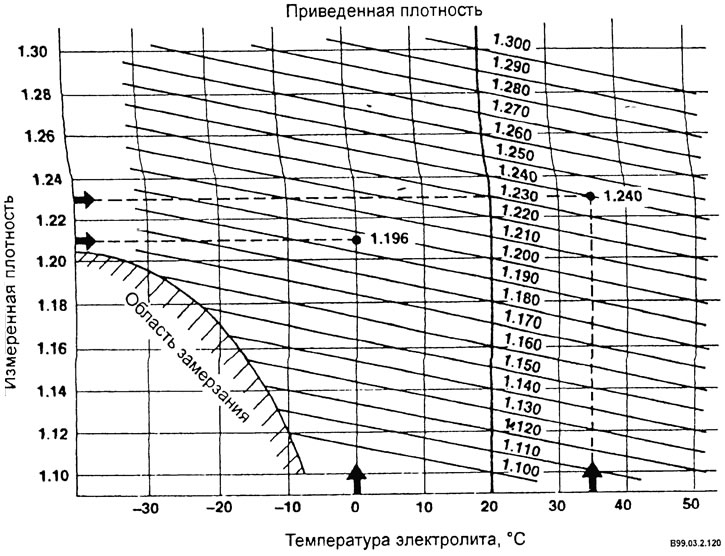
There should be very little difference between different banks, and if the difference exceeds 0.025 g/cm3, then the reasons for this may be the following:
- loss of electrolyte from the battery caused by splashing or leakage which can lead to a decrease in the density of the electrolyte when the lack of electrolyte is compensated by topping up with distilled water instead of fresh electrolyte;
- an internal short circuit, caused by bending of the plates or something like that, leads to a malfunction of the entire battery and its inevitable rapid failure. The density of the electrolyte at various temperatures for a fully charged battery is shown in Table A. The density of the electrolyte for a fully discharged battery at various temperatures is shown in Table B.
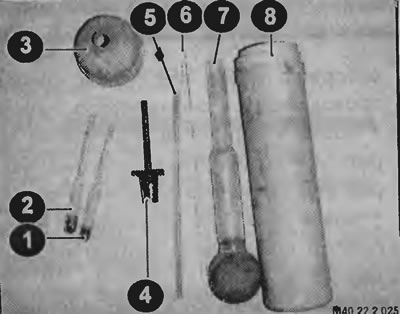
Elements of the device for checking the correction of the density of the electrolyte of the battery and the density of the coolant (antifreeze)
1 - hydrometer for checking the density of the battery electrolyte; 2 - hydrometer for checking the density of the coolant; 3 - funnel; 4 - extension; 5 - glass tube; 6 - glass rod; 7 - container with a rubber pear; 8 - case.
Table A. Electrolyte density (battery is fully charged)
| Density, g/cm3 | Temperature,°С |
| 1,268 | 38 |
| 1,272 | 32 |
| 1,276 | 27 |
| 1,280 | 21 |
| 1,284 | 16 |
| 1,288 | 10 |
| 1,292 | 4 |
| 1,296 | -1 5 |
Table B. Electrolyte density (the battery is completely discharged)
| Density, g/cm3 | Temperature,°С |
| 1,098 | 38 |
| 1,102 | 32 |
| 1,106 | 27 |
| 1,110 | 21 |
| 1,114 | 16 |
| 1,118 | 10 |
| 1,122 | 4 |
| 1,126 | -1,5 |
